
The element was discovered in 1827 by F. Woehler. The name comes from the Latin alumen (alum). The element is also called aluminium.
Ionization energies
AlI 6.0 eV, AlII 18.8 eV, AlIII 28.4 eV, AlIV 120 eV.
Absorption lines of AlI
| W(3944) | (resonance line | ||||
| UV M.1) | W(6699)(5) | ||||
| Group | V | Ib | V | III | Ib |
| B 7 | 0.006 | ||||
| B 9 | 0.051 | ||||
| A 0 | 0.085 | 0.05 | |||
| A 1 | 0.098 | ||||
| A 2 | 0.14 | 0.085(Ia) | |||
| A 3 | 0.050(0) | ||||
| A 7 | 0.18 | ||||
| F 0 | 0.30(Ia) | ||||
| F 4 | 0.29 | ||||
| F 5 | 0.015 | ||||
| F 6 | 0.47 | ||||
| F 8 | 0.35 | 0.28 | |||
| G 1 | 0.4 | 0.056 | |||
| G 2 | 0.50 | 0.018 | 0.073, 0.092 | ||
| S | 0.488 | 0.021 | |||
| G 5 | 0.093 | ||||
| G 6 | 0.142 | ||||
| G 8 | 0.022(IV) | 0.119 | |||
| K 0 | 0.006 | 0.052 | |||
| K 2 | 0.062 | 0.188 | |||
| K 3 | 0.11 | ||||
| K 5 | 0.063 | 0.157 | |||
| M 2 | 0.117 | ||||
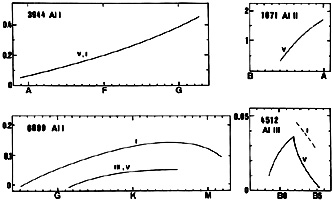
AlI (for instance the line at 3944) is present in dwarfs from late B-type stars onwards, increasing steadily in strength toward G-type stars. This line shows no luminosity effect. The line at 6699 shows a positive luminosity effect from late F-type onwards.
Emission lines of AlI
That at 3944 (1) appears in emission from some T Tau stars (Joy 1945)
Absorption lines of AlII
AlII has its resonance line at 1671 (UV M.2). Sadakane et al. (1983) provide the following values.
| Group | V |
| B 5 | 0.59(IV) |
| B 7 | 1.02 |
| B 9 | 1.60 |
The ultraviolet blend at 1723(6) shows a positive luminosity effect in B-type stars (Heck et al. 1984).
In the sun a line at 3900.66 is probably AlII
Al III is present in B-type and more weakly in A-type stars.
Absorption lines of AlIII
AlIII has its resonance lines in the ultraviolet (1854 and 1863 - UV M.1). Sadakane et al. (1983) provide the following values.
| W(1863)(1) | W(3612)(1) | W(4512)(3) | |||
| Group | V | V | V | III | Ia |
| 0 7 | 0.004 | ||||
| 0 9 | 0.015 | ||||
| B 0 | 0.012 | 0.022 | |||
| B 0.5 | 0.032 | 0.030 | |||
| B 2 | 0.027(IV) | 0.036 | 0.046 | ||
| B 3 | 0.012 | 0.017 | 0.031 | ||
| B 5 | 0.48(IV) | 0.004 | 0.039 | ||
| B 7 | 0.40 | ||||
| B 9 | 0.20 | ||||
AlIII (for instance the line at 4512) is present in B-type stars with a maximum around B5 and a positive luminosity effect. AlIII 4149(5) is present in the solar chromosphere, besides AlI and AlII (Pierce 1968).
 |
Behavior in non-normal stars
Sadakane et al. (1983) found that both AlII and AlIII are weak in Bp stars of the Hg-Mn, Si and He-weak subgroups. The extent of weakening varies from star to star, with equivalent widths lesser by factors two to six than in normal stars.
AlI is normal or slightly weak in Ap stars of the Cr-EU-Sr type (Adelman 1973b).
Al is either slightly strengthened in Am stars (Burkhart and Coupry 1991) or normal (Smith 1973, 1974).
Al is probably underabundant with respect to Fe in the most metal-weak stars (Fe/H × 10-4) by factors of the order of 4-5 (Molaro and Bonifacio 1990). This is also true for horizontal branch stars (Adelman and Philip 1990, 1992a). Magain (1989) suggests that Al has a normal (i.e. solar) abundance in normal stars, whereas it is increasingly deficient in increasingly metal-weak dwarfs, a fact that was also pointed out by Gratton and Sneden (1988). Since not all authors use the same lines, the abundances differ between authors more than would seem reasonable (Spite 1992).
Al apparently has an erratic behavior from star to star in globular cluster stars, but seems to be overabundant with respect to iron (Francois 1991). However, Cionzalez and Wallerstein (1992) find it underabundant in one F-type globular cluster supergiant.
In novae AlII and AlIII lines usually appear in emission in the ultraviolet region during the `principal spectrum' phase. Lines of [AlIV] and [AlVIII] sometimes appear during the nebular stage (Warner 1989).
Al is very overabundant in the spectra of novae of the O-Ne-Mg subgroup (Andreae 1993).
Isotopes
Al has eight isotopes and isomers. The only stable one is Al27. One of the isotopes, Al26, has a half life of 7.4 × 105 years. Branch and Peery (1970) derive Al26 / Al27 < 0.15 from the AlH bands in one S-qpe star.
Origin
The element can be produced by neon burning or by explosive nucleosynthesis.
Published in "The Behavior of Chemical Elements in Stars", Carlos Jaschek and Mercedes Jaschek, 1995, Cambridge University Press.