
This element has been known since prehistory. The name comes from the Latin word Cuprum (Cyprus), because on this island there existed heavily exploited copper mines.
Ionization energies
CuI 7.7 eV, CuII 20.3 eV, CuIII 36.8 eV.
Absorption lines of CuI
| Group | V | III | Ib |
| F0 | 0.035 | ||
| F4 | 0.025 | ||
| F5 | 0.049 | 0.030 | |
| F6 | 0.037 | ||
| F8 | 0.037 | 0.083 | |
| G0 | 0.060 | 0.158 | |
| G1 | 0.083 | 0.184 | |
| S | 0.082 | 0.203 | |
| G2 | 0.092 | 0.178 | |
| G5 | 0.245 | ||
| G6 | 0.301 | ||
| G8 | 0.112(IV) | 0.15 | 0.286 |
| K0 | 0.110 | 0.200 | |
| K2 | 0.182 | 0.269,0.354 | |
| K3 | 0.230 | ||
| K5 | 0.309,0.368,0.394 | ||
| M0 | 0.58 | ||
| M2 | 0.388,0.58(Ia) | ||
| M3 | 0.34 | ||
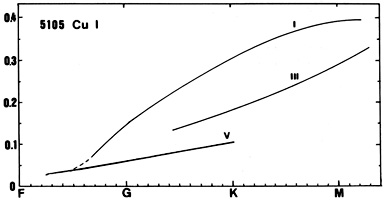
Cu I (see the line at 5105) appears in early F-type stars and intensifies toward later types. A positive luminosity effect exists.
Absorption lines of CuII
CuII lines are visible in the ultraviolet spectral region of B- and A-type
stars.
Emission lines of CuII
The 3806 line from [CuII] is present in the luminous blue variable
eta Car (Thackeray 1953) and in one typical B[e] star (Swings
1973).
Behavior in non-normal stars
The first detection of the resonance line of CuII 1358(M.3) line in Ap
stars was made by Bidelman (1979). Jacobs and Dworetsky (1981)
measured the line in several Bp stars of the Hg-Mn subgroup and
found it to be very strong (W up to 0.270). Takada-Hidai (1991)
noticed, however, that in some
stars of this subgroup Cu can be very weak or absent.
Cu I lines are also strong in Am stars. Typical W values are twice as large as in normal stars of the same temperature (Smith 1973, 1974).
Sneden et al. (1991) find that, in metal-weak (disk and halo) stars, Cu is weakened with respect to the metals Fe and Ni, to the point that, in extreme metal-weak objects, Cu lines are undetectable. A similar behavior is found in globular cluster stars (Wheeler et al. 1989). One emission line of [Cu II] is seen in one VV Cep star (Rossi et al. 1992).
Isotopes
Cu has nine short-lived isotopes and two stable ones, Cu63 and
Cu65. In the solar system 69% of all Cu is in the form of
Cu63 and the remainder is in the form of Cu65.
Origin
Cu63 and Cu65 are both produced by explosive
nucleosynthesis, but Cu63 can also be produced by carbon burning.
Published in "The Behavior of Chemical Elements in Stars", Carlos Jaschek and Mercedes Jaschek, 1995, Cambridge University Press.