
This element was isolatod in 1781 by P. Hjelm in Uppsala, Sweden. The name alludes to molybdos, which in Greek means lead, an element for which it was mistaken.
Ionization energies
MoI 7.1 eV, MoII 16.1 eV, MoIII 27.2 eV.
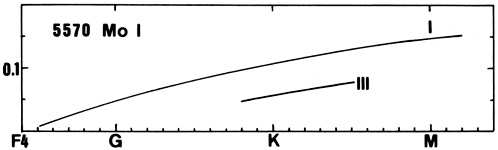
| 5506(4) | 5570(4) | ||||
| Group | V | III | V | III | Ib |
| F5 | 0.016 | ||||
| G2 | 0.004 | ||||
| S | 0.005 | 0.006 | |||
| G9 | 0.022 | ||||
| K0 | 0.044 | 0.034 | |||
| K2 | 0.069 | 0.100 | |||
| K3 | 0.088 | ||||
| K5 | 0.120 | ||||
| M2 | 0.151 | ||||
MoI has many faint lines in the solar spectrum and that of later type stars. It increases in strength toward cooler stars (see 5570) and shows a positive luminosity effect.
Absorption lines of MoII
The equivalent width of MoII 3320(6) in the sun is 0.009. MoII lines are
also present in the ultraviolet at 2038(32) and 2045(22) (Sadakane
1991).
 |
Behavior in non-normal stars
Jaschek and Brandi (1972) and Cowley et al. (1974) detected Mo II in two
Ap stars of the Cr-Eu-Sr subgroup. Adelman (1973b) signaled the
presence of Mo in more stars of this subgroup.
MoI lines are enhanced in the spectra of Ba stars, which leads to overabundances of one order of magnitude (Lambert 1985, Smith 1984). Typical W values are twice as large as in normal giants (Danziger 1965).
Mo I lines are enhanced in S-type stars, which leads to overabundances of one order of magnitude (Smith and Wallerstein 1983) and in SC stars (Kipper and Wallerstein 1990).
Isotopes
Mo has seven stable isotopes and 14 short-lived isotopes and isomers. The
stable isotopes are Mo 92, 94, 95, 96, 97, 98 and 100, which occur in
the solar system with frequencies 15%, 9%, 16%, 17%, 10%, 24% and 9%
respectively.
Origin
Mo92and Mo94 can only be produced by the p
process; Mo 95, 97 and 98 are produced by either the r or the s
process. Mo96 is a pure s process and Mo100 a pure
r process product.
Published in "The Behavior of Chemical Elements in Stars", Carlos Jaschek and Mercedes Jaschek, 1995, Cambridge University Press.