
This element was discovered by A. del Rio in Mexico City, Mexico, in 1801 and rediscovered by G. Selfström in Sweden in 1831. The name comes from the Scandinavian goddess Vanadis.
Ionization energies
VI6.7eV,VII 14.6eV,VIII29.3 eV,VIV51.4eV,VV71.8eV,VVI 141 eV,VVII
165 eV, VVIII 191 eV.
Many faint lines of V are present in stellar spectra. In the sun, V figures in fifth place among the elements as far as the number of lines is concerned.
Absorption lines of VI
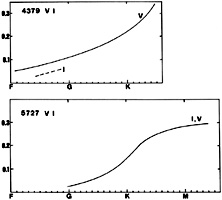
| 4379(22) | 5727(35) | ||||
| Group | V | Ib | V | III | Ib |
| F0 | 0.06 | 0.026(II) | |||
| F4 | 0.012 | ||||
| F5 | 0.10,0.04 | 0.024 | |||
| F6 | 0.023 | ||||
| F8 | 0.070 | 0.013 | |||
| G0 | 0.11 | 0.014 | |||
| G1 | 0.043 | 0.018 | |||
| G2 | 0.11 | 0.035 | 0.035 | ||
| S | 0.110 | 0.037 | |||
| G5 | 0.15 | 0.047 | 0.054 | ||
| G8 | 0.037(IV) | 0.107 | 0.098 | ||
| K0 | 0.24 | 0.123,0.170 | |||
| K2 | 0.163 | 0.204 | |||
| K3 | 0.213 | 0.230 | |||
| K5 | 0.34 | 0.263 | |||
| M0 | 0.281 | ||||
| M2 | 0.269 | ||||
| M2.5 | 0.288 | ||||
VI (for instance the lines at 4379 and 5727) appears in late A-type stars and increases toward later spectral types. A slight negative luminosity effect before type G may be present in the 4379 line.
 |
Emission lines of Vl
VI lines from M.34 and 48 were observed in emission in one
Mira-type variable by Grudzinska (1984).
Absorption lines of Vll
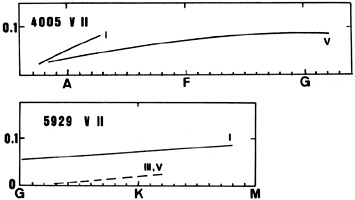
| 4005(32) | 5929(98) | ||||
| Group | V | Ia | V | III | Ib |
| B8 | 0.017 | ||||
| B9 | 0.024 | ||||
| A0 | 0.02 | 0.03,0.050 | |||
| A1 | 0.061 | ||||
| A2 | 0.072,0.061 | 0.069(Ia) | |||
| A3 | 0.115(0) | ||||
| A7 | 0.045 | ||||
| G0 | 0.039 | ||||
| G1 | 0.055 | ||||
| G2 | 0.047 | ||||
| S | 0.085 | 0.004 | |||
| G5 | 0.062 | ||||
| G8 | 0.052 | ||||
| K2 | 0.033 | 0.073 | |||
| K3 | 0.089 | ||||
| K5 | 0.064,0.101 | ||||
VII (for instance the lines at 4005 and 5929) appears in late B-type stars and increases toward later types. A positive luminosity effect exists.
 |
Forbidden lines
VIII forbidden lines are observed in one recurrent nova Joy and
Swings 1945).
Behavior in non-normal stars
V seems to behave in a manner parallel to that of Fe over the whole range of
Fe abundances, and it can thus be regarded as a 'typical metal'. In
globular cluster stars, V seems to be slightly overabundant with
respect to iron by factors of the order of two (Wheeler et al. 1989).
Isotopes
V has two stable isotopes, V50 and V51. In the
solar system
they occur with 99.75% and 0.25% abundances. There exist also seven
short-lived isotopes. The isotopic effect in the VO bands due to the two
stable isotopes would be very small and apparently no search has been
undertaken.
Origin
V50 and V51 are produced by explosive
nucleosynthesis, but V50 can also be produced by the nuclear
statistical equilibrium process.
Published in "The Behavior of Chemical Elements in Stars", Carlos Jaschek and Mercedes Jaschek, 1995, Cambridge University Press.