


6.1. Continuum versus line absorption
X-rays emitted by cosmic sources do not travel unattenuated to a distant
observer. This is because intervening matter in the line of sight
absorbs a part of the X-rays. With low-resolution instruments,
absorption can be studied only through the measurement of broad-band
flux depressions caused by continuum absorption. However, at high
spectral resolution also absorption lines can be studied, and in fact
absorption lines offer more sensitive tools to detect weak intervening
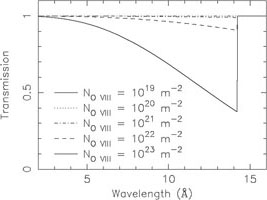
absorption systems. We illustrate this in Fig. 9.
At an O VIII column density of
1021 m-2, the absorption edge has an optical
depth of 1%; for the same column density, the core of the
Ly line is already saturated
and even for 100 times lower column density, the line core still has an
optical depth of 5%.
line is already saturated
and even for 100 times lower column density, the line core still has an
optical depth of 5%.
 |
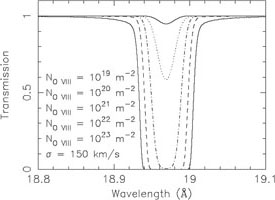 |
Figure 9. Continuum (left) and
Ly |
|
Continuum absorption can be calculated simply from the photoionisation
cross sections, that we discussed already in
Sect. 3.2.2. The total continuum
opacity  cont can
be written as
cont can
be written as
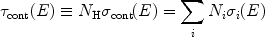 |
(41) |
i.e, by averaging over the various ions i with column density
Ni. Accordingly, the continuum transmission
T(E) of such a clump of matter can be written as
T(E) =
exp(- cont(E)).
For a worked out example see also Sect. 6.6.
cont(E)).
For a worked out example see also Sect. 6.6.
When light from a background source shines through a clump of matter, a
part of the radiation can be absorbed. We discussed already the
continuum absorption. The transmission in a spectral line at wavelength
 is given by
is given by
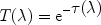 |
(42) |
with
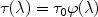 |
(43) |
where 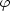 (
( ) is the line profile and
) is the line profile and
 0 is the opacity
at the line centre
0 is the opacity
at the line centre
 0, given by:
0, given by:
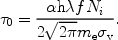 |
(44) |
Apart from the fine structure constant
 and Planck's constant
h, the optical depth also depends on the properties of the
absorber, namely the ionic column density Ni and the
velocity dispersion
and Planck's constant
h, the optical depth also depends on the properties of the
absorber, namely the ionic column density Ni and the
velocity dispersion
 v.
Furthermore, it depends on the oscillator strength f which is a
dimensionless quantity that is different for each transition and is of
order unity for the strongest transitions.
v.
Furthermore, it depends on the oscillator strength f which is a
dimensionless quantity that is different for each transition and is of
order unity for the strongest transitions.
In the simplest approximation, a Gaussian profile
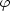 (
( ) = exp
-(
) = exp
-( -
-
 0)2
/ b2) can be adopted, corresponding to pure Doppler
broadening for a thermal plasma. Here b = 21/2
0)2
/ b2) can be adopted, corresponding to pure Doppler
broadening for a thermal plasma. Here b = 21/2
 with
with
 the normal
Gaussian root-mean-square width. The full width at half maximum of this
profile is given by (ln 256)1/2
the normal
Gaussian root-mean-square width. The full width at half maximum of this
profile is given by (ln 256)1/2
 or approximately
2.35
or approximately
2.35 . One may even
include a turbulent velocity
. One may even
include a turbulent velocity
 t into the
velocity width
t into the
velocity width
 v, such that
v, such that
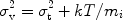 |
(45) |
with mi the mass of the ion (we have tacitly changed
here from wavelength to velocity units through the scaling
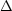
 /
/
 0
=
0
= 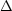 v / c).
v / c).
The equivalent width W of the line is calculated from
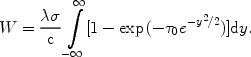 |
(46) |
For the simple case that
 0 << 1,
the integral can be evaluated as
0 << 1,
the integral can be evaluated as
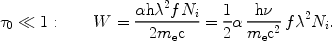 |
(47) |
A Gaussian line profile is only a good approximation when the Doppler
width of the line is larger than the natural width of the line. The
natural line profile for an absorption line is a Lorentz profile
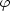 (
( ) = 1 / (1 +
x2) with x =
4
) = 1 / (1 +
x2) with x =
4
 / A. Here
/ A. Here
 is the frequency difference
is the frequency difference
 -
-
 0 and A is
the total transition probability from the upper level downwards to any
level, including all radiative and Auger decay channels.
0 and A is
the total transition probability from the upper level downwards to any
level, including all radiative and Auger decay channels.
Convolving the intrinsic Lorentz profile with the Gaussian Doppler profile due to the thermal or turbulent motion of the plasma, gives the well-known Voigt line profile
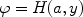 |
(48) |
where
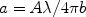 |
(49) |
and y = c
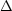
 / b
/ b . The
dimensionless parameter a (not to be
confused with the a in Eqn. 4) represents the relative
importance of the Lorentzian term (~ A) to the Gaussian term (~
b). It should be noted here that formally for a > 0 the
parameter
. The
dimensionless parameter a (not to be
confused with the a in Eqn. 4) represents the relative
importance of the Lorentzian term (~ A) to the Gaussian term (~
b). It should be noted here that formally for a > 0 the
parameter  0
defined by
(44) is not exactly the optical depth at line centre, but as long as
a << 1 it is a fair approximation. For a >> 1
it can be shown that H(a, 0)
0
defined by
(44) is not exactly the optical depth at line centre, but as long as
a << 1 it is a fair approximation. For a >> 1
it can be shown that H(a, 0)
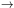 1 /
a
1 /
a 1/2.
1/2.
6.4. Some important X-ray absorption lines
There are several types of absorption lines. The most well-known are the
normal strong resonance lines, which involve electrons from the
outermost atomic shells. Examples are the 1s-2p line of O VII at
21.60 Å, the O VIII
Ly doublet at 18.97
Å and the well known 2s-2p
doublet of O VI at 1032 and 1038 Å. See
Richter et
al. 2008
- Chapter 3, this volume, for an extensive discussion on these
absorption lines in the WHIM.
doublet at 18.97
Å and the well known 2s-2p
doublet of O VI at 1032 and 1038 Å. See
Richter et
al. 2008
- Chapter 3, this volume, for an extensive discussion on these
absorption lines in the WHIM.
The other class are the inner-shell absorption lines. In this case the excited atom is often in an unstable state, with a large probability for the Auger process (Sect. 3.2.4). As a result, the parameter a entering the Voigt profile (Eqn. 49) is large and therefore the lines can have strong damping wings.
In some cases the lines are isolated and well resolved, like the O VII and O VIII 1s-2p lines mentioned above. However, in other cases the lines may be strongly blended, like for the higher principal quantum number transitions of any ion in case of high column densities. Another important group of strongly blended lines are the inner-shell transitions of heavier elements like iron. They form so-called unresolved transition arrays (UTAs); the individual lines are no longer recognisable. The first detection of these UTAs in astrophysical sources was in a quasar (Sako et al., 2001).
In Table 6 we list the 70 most important X-ray
absorption lines for  < 100 Å. The importance was
determined by calculating the maximum ratio W /
< 100 Å. The importance was
determined by calculating the maximum ratio W /
 that these
lines reach for any given temperature.
that these
lines reach for any given temperature.
 |
ion | Wmax | log |  |
ion | Wmax | log | |
| (Å) | (mÅ) | Tmax | (Å) | (mÅ) | Tmax | |||
| 9.169 | Mg XI | 1.7 | 6.52 | 31.287 | N I | 5.5 | 4.04 | |
| 13.447 | Ne IX | 4.6 | 6.38 | 33.426 | C V | 7.0 | 5.76 | |
| 13.814 | Ne VII | 2.2 | 5.72 | 33.734 | C VI | 12.9 | 6.05 | |
| 15.014 | Fe XVII | 5.4 | 6.67 | 33.740 | C VI | 10.3 | 6.03 | |
| 15.265 | Fe XVII | 2.2 | 6.63 | 34.973 | C V | 10.3 | 5.82 | |
| 15.316 | Fe XV | 2.3 | 6.32 | 39.240 | S XI | 6.5 | 6.25 | |
| 16.006 | O VIII | 2.6 | 6.37 | 40.268 | C V | 17.0 | 5.89 | |
| 16.510 | Fe IX | 5.6 | 5.81 | 40.940 | C IV | 7.2 | 5.02 | |
| 16.773 | Fe IX | 3.0 | 5.81 | 41.420 | C IV | 14.9 | 5.03 | |
| 17.396 | O VII | 2.8 | 6.06 | 42.543 | S X | 6.3 | 6.14 | |
| 17.768 | O VII | 4.4 | 6.11 | 50.524 | Si X | 11.0 | 6.14 | |
| 18.629 | O VII | 6.9 | 6.19 | 51.807 | S VII | 7.6 | 5.68 | |
| 18.967 | O VIII | 8.7 | 6.41 | 55.305 | Si IX | 14.0 | 6.06 | |
| 18.973 | O VIII | 6.5 | 6.39 | 60.161 | S VII | 12.2 | 5.71 | |
| 19.924 | O V | 3.2 | 5.41 | 61.019 | Si VIII | 11.9 | 5.92 | |
| 21.602 | O VII | 12.1 | 6.27 | 61.070 | Si VIII | 13.2 | 5.93 | |
| 22.006 | O VI | 5.1 | 5.48 | 62.751 | Mg IX | 11.1 | 5.99 | |
| 22.008 | O VI | 4.1 | 5.48 | 68.148 | Si VII | 10.1 | 5.78 | |
| 22.370 | O V | 5.4 | 5.41 | 69.664 | Si VII | 10.9 | 5.78 | |
| 22.571 | O IV | 3.6 | 5.22 | 70.027 | Si VII | 11.3 | 5.78 | |
| 22.739 | O IV | 5.1 | 5.24 | 73.123 | Si VII | 10.7 | 5.78 | |
| 22.741 | O IV | 7.2 | 5.23 | 74.858 | Mg VIII | 17.1 | 5.93 | |
| 22.777 | O IV | 13.8 | 5.22 | 80.449 | Si VI | 13.0 | 5.62 | |
| 22.978 | O III | 9.4 | 4.95 | 80.577 | Si VI | 12.7 | 5.61 | |
| 23.049 | O III | 7.1 | 4.96 | 82.430 | Fe IX | 14.0 | 5.91 | |
| 23.109 | O III | 15.1 | 4.95 | 83.128 | Si VI | 14.2 | 5.63 | |
| 23.350 | O II | 7.5 | 4.48 | 83.511 | Mg VII | 14.2 | 5.82 | |
| 23.351 | O II | 12.6 | 4.48 | 83.910 | Mg VII | 19.8 | 5.85 | |
| 23.352 | O II | 16.5 | 4.48 | 88.079 | Ne VIII | 15.2 | 5.80 | |
| 23.510 | O I | 7.2 | 4.00 | 95.385 | Mg VI | 15.5 | 5.67 | |
| 23.511 | O I | 15.0 | 4.00 | 95.421 | Mg VI | 18.7 | 5.69 | |
| 24.779 | N VII | 4.6 | 6.20 | 95.483 | Mg VI | 20.5 | 5.70 | |
| 24.900 | N VI | 4.0 | 5.90 | 96.440 | Si V | 16.8 | 5.33 | |
| 28.465 | C VI | 4.6 | 6.01 | 97.495 | Ne VII | 22.8 | 5.74 | |
| 28.787 | N VI | 9.8 | 6.01 | 98.131 | Ne VI | 16.7 | 5.65 | |
Note the dominance of lines from all ionisation stages of oxygen in the 17-24 Å band. Furthermore, the Fe IX and Fe XVII lines are the strongest iron features. These lines are weaker than the oxygen lines because of the lower abundance of iron; they are stronger than their neighbouring iron ions because of somewhat higher oscillator strengths and a relatively higher ion fraction (cf. Fig. 7).
Note that the strength of these lines can depend on several other parameters, like turbulent broadening, deviations from CIE, saturation for higher column densities, etc., so some care should be taken when the strongest line for other physical conditions are sought.
In most practical situations, X-ray absorption lines are unresolved or poorly resolved. As a consequence, the physical information about the source can only be retrieved from basic parameters such as the line centroid (for Doppler shifts) and equivalent width W (as a measure of the line strength).
As outlined in Sect. 6.3, the
equivalent width W of a spectral line from an ion i
depends, apart from atomic parameters, only on the column density
Ni and the velocity broadening
 v. A
useful diagram is the so-called curve of growth. Here we present it in
the form of a curve giving W versus Ni for a
fixed value of
v. A
useful diagram is the so-called curve of growth. Here we present it in
the form of a curve giving W versus Ni for a
fixed value of  v.
v.
The curve of growth has three regimes. For low column densities, the
optical depth at line centre
 0 is small, and
W increases linearly with Ni, independent of
0 is small, and
W increases linearly with Ni, independent of
 v (Eqn. 47). For
increasing column density,
v (Eqn. 47). For
increasing column density,
 0 starts to
exceed unity and then W increases only logarithmically with
Ni. Where this happens exactly, depends on
0 starts to
exceed unity and then W increases only logarithmically with
Ni. Where this happens exactly, depends on
 v. In this
intermediate logarithmic branch W depends both on
v. In this
intermediate logarithmic branch W depends both on
 v and
Ni, and hence
by comparing the measured W with other lines from the same ion,
both parameters can be estimated. At even higher column densities, the
effective opacity increases faster (proportional to
(Ni)1/2), because of the
Lorentzian line wings. Where this happens depends on the Voigt parameter
a (Eqn. 49). In this range of
densities, W does not depend any more on
v and
Ni, and hence
by comparing the measured W with other lines from the same ion,
both parameters can be estimated. At even higher column densities, the
effective opacity increases faster (proportional to
(Ni)1/2), because of the
Lorentzian line wings. Where this happens depends on the Voigt parameter
a (Eqn. 49). In this range of
densities, W does not depend any more on
 v.
v.
In Fig. 10 we show a few characteristic
examples. The examples of O I and O VI illustrate the higher W
for UV lines as compared to X-ray lines (cf. Eqn. 47) as well as the
fact that inner shell
transitions (in this case the X-ray lines) have larger a-values
and hence reach sooner the square root branch. The example of O VII
shows how different lines from the same ion have different equivalent
width ratios depending on
 v, hence offer an
opportunity to determine that parameter. The last frame shows the
(non)similarties for similar transitions in different ions. The
innershell nature of the transition in O VI yields a higher a
value and hence an earlier onset of the square root branch.
v, hence offer an
opportunity to determine that parameter. The last frame shows the
(non)similarties for similar transitions in different ions. The
innershell nature of the transition in O VI yields a higher a
value and hence an earlier onset of the square root branch.
6.6. Galactic foreground absorption
All radiation from X-ray sources external to our own Galaxy has to pass
through the interstellar medium of our Galaxy, and the intensity is
reduced by a factor of e -
 (E) with the
optical depth
(E) with the
optical depth  (E) =
(E) =
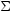 i
i
 i(E)
i(E)
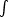 ni(
ni(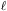 )
d
)
d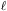 , with the summation
over all relevant ions i and the integration over the line of sight
d
, with the summation
over all relevant ions i and the integration over the line of sight
d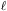 . The absorption
cross section
. The absorption
cross section
 i(E)
is often taken to be simply
the (continuum) photoionisation cross section, but for high-resolution
spectra it is necessary to include also the line opacity, and for very
large column densities also other processes such as Compton ionisation
or Thomson scattering.
i(E)
is often taken to be simply
the (continuum) photoionisation cross section, but for high-resolution
spectra it is necessary to include also the line opacity, and for very
large column densities also other processes such as Compton ionisation
or Thomson scattering.
For a cool, neutral plasma with cosmic abundances one often writes
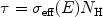 |
(50) |
where the hydrogen column density NH

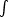 nH dx. In
nH dx. In
 eff(E)
all contributions to the absorption from all elements as well as their
abundances are taken into account.
eff(E)
all contributions to the absorption from all elements as well as their
abundances are taken into account.
The relative contribution of the elements is also made clear in Fig. 11. Below 0.28 keV (the carbon edge) hydrogen and helium dominate, while above 0.5 keV in particular oxygen is important. At the highest energies, above 7.1 keV, iron is the main opacity source.
Yet another way to represent these data is to plot the column density or distance for which the optical depth becomes unity (Fig. 12). This figure shows that in particular at the lowest energies X-rays are most strongly absorbed. The visibility in that region is thus very limited. Below 0.2 keV it is extremely hard to look outside our Milky Way.
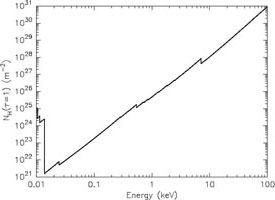 |
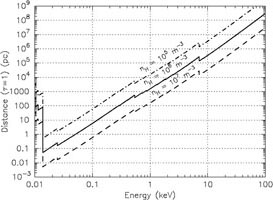 |
Figure 12. Left panel: Column
density for which the optical depth becomes unity. Right panel:
Distance for which |
|
The interstellar medium is by no means a homogeneous, neutral, atomic gas. In fact, it is a collection of regions all with different physical state and composition. This affects its X-ray opacity. We briefly discuss here some of the most important features.
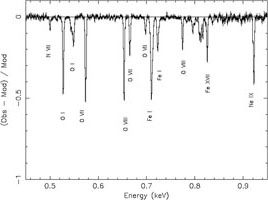
The ISM contains cold gas (< 50 K), warm, neutral or lowly ionised gas (6000 - 10000 K) as well as hotter gas (a few million K). We have seen before (see Sect. 3.2.2) that for ions the absorption edges shift to higher energies for higher ionisation. Thus, with instruments of sufficient spectral resolution, the degree of ionisation of the ISM can be deduced from the relative intensities of the absorption edges. Cases with such high column densities are rare, however (and only occur in some AGN outflows), and for the bulk of the ISM the column density of the ionised ISM is low enough that only the narrow absorption lines are visible (see Fig. 9). These lines are only visible when high spectral resolution is used (see Fig. 13). It is important to recognise these lines, as they should not be confused with absorption line from within the source itself. Fortunately, with high spectral resolution the cosmologically redshifted absorption lines from the X-ray source are separated well from the foreground hot ISM absorption lines. Only for lines from the Local Group this is not possible, and the situation is here complicated as the expected temperature range for the diffuse gas within the Local Group is similar to the temperature of the hot ISM.
 |
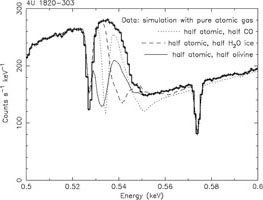 |
Figure 13. Left panel: Simulated
100 ks absorption spectrum as observed with the
Explorer of Diffuse Emission and Gamma-ray Burst Explosions (EDGE), a
mission proposed for ESA's Cosmic Vision program. The parameters of the
simulated source are similar to those of 4U 1820-303
(Yao & Wang,
2006).
The plot shows the residuals of the simulated spectrum if the
absorption lines in the model are ignored. Several characteristic
absorption features of both neutral and ionised gas are indicated. |
|
Another ISM component is dust. A significant fraction of some atoms can be bound in dust grains with varying sizes, as shown below (from Wilms et al. 2000):
| H | He | C | N | O | Ne | Mg | Si | S | Ar | Ca | Fe | Ni |
| 0 | 0 | 0.5 | 0 | 0.4 | 0 | 0.8 | 0.9 | 0.4 | 0 | 0.997 | 0.7 | 0.96 |
The numbers represent the fraction of the atoms that are bound in dust grains. Noble gases like Ne and Ar are chemically inert hence are generally not bound in dust grains, but other elements like Ca exist predominantly in dust grains. Dust has significantly different X-ray properties compared to gas or hot plasma. First, due to the chemical binding, energy levels are broadened significantly or even absent. For example, for oxygen in most bound forms (like H2O) the remaining two vacancies in the 2p shell are effectively filled by the two electrons from the other bound atom(s) in the molecule. Therefore, the strong 1s-2p absorption line at 23.51 Å (527 eV) is not allowed when the oxygen is bound in dust or molecules, because there is no vacancy in the 2p shell. Transitions to higher shells such as the 3p shell are possible, however, but these are blurred significantly and often shifted due to the interactions in the molecule. Each constituent has its own fine structure near the K-edge (Fig. 13b). This fine structure offers therefore the opportunity to study the (true) chemical composition of the dust, but it should be said that the details of the edges in different important compounds are not always (accurately) known, and can differ depending on the state: for example water, crystalline and amorphous ice all have different characteristics. On the other hand, the large scale edge structure, in particular when observed at low spectral resolution, is not so much affected. For sufficiently high column densities of dust, self-shielding within the grains should be taken into account, and this reduces the average opacity per atom.
Finally, we mention here that dust also causes scattering of X-rays. This is in particular important for higher column densities. For example, for the Crab nebula (NH = 3.2 × 1025 m-2), at an energy of 1 keV about 10% of all photons are scattered in a halo, of which the widest tails have been observed out to a radius of at least half a degree; the scattered fraction increases with increasing wavelength.